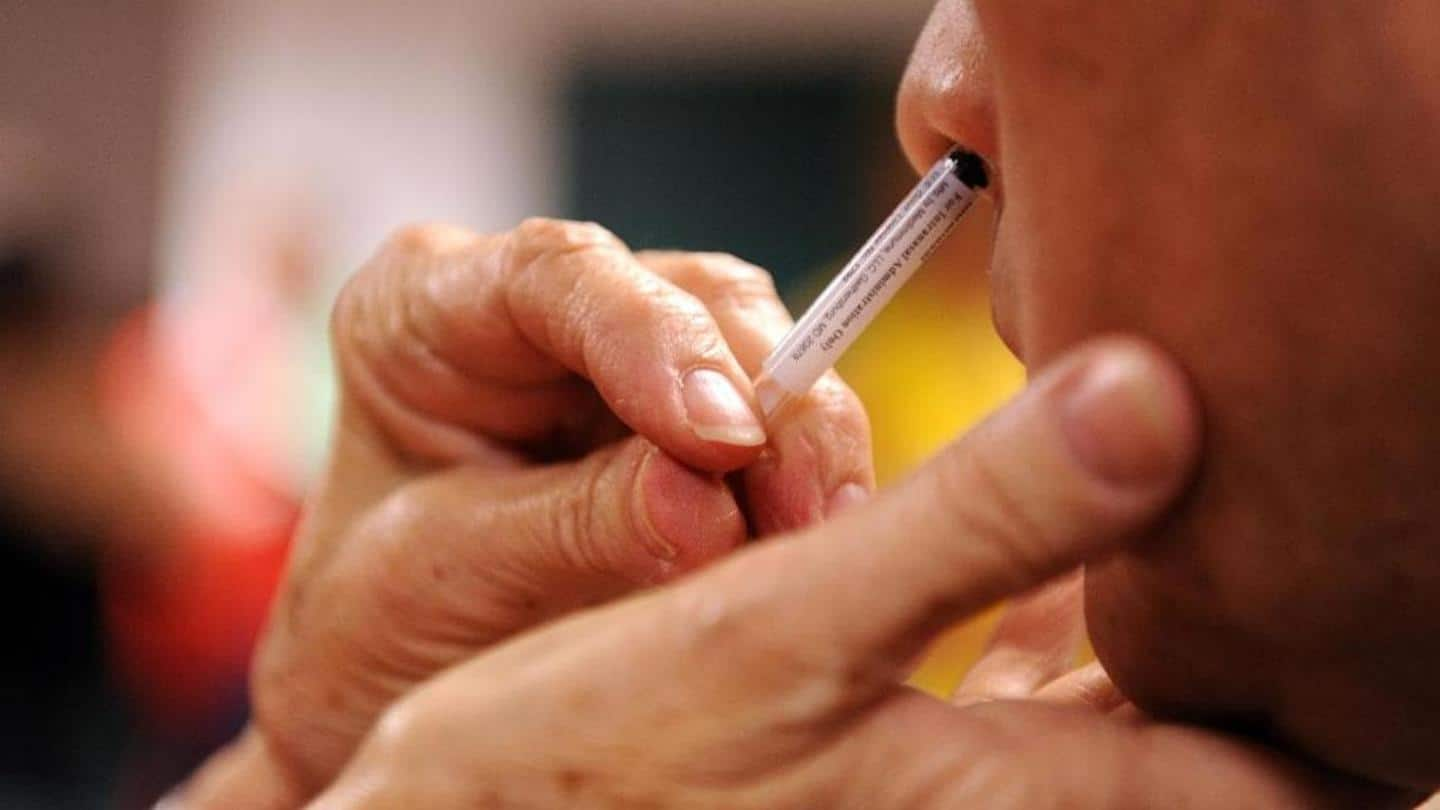
Bharat Biotech signs deal for nasal COVID-19 vaccine: Details here
What's the story
In a major move, Bharat Biotech has signed an agreement to license a nasal COVID-19 vaccine developed by the Washington University School of Medicine in St. Louis, United States. The agreement will see the Indian biotech firm manufacture and distribute the novel solution for the deadly respiratory disease across several countries, including our own. Here is all you need to know about the agreement.
Deal
What does the agreement entail?
Under the deal, announced just recently, Bharat Biotech and US-based biotech firm Precision Virologics acquired rights to WUSM's chimp-adenovirus intra-nasal vaccine for COVID-19. The US firm has access to the vaccine technology for supply in the US, Europe, and Japan, while the Indian company will be handling distribution in all other markets, starting with India.
Details
Bharat Biotech will conduct clinical trials in India
The single dose nasal vaccine has already shown "unprecedented levels of protection in mouse studies" and is cleared for a Phase-1 human trial, which will be conducted at Saint Louis University's Vaccine and Treatment Evaluation Unit. The advanced phases of the clinical study will be carried out in India upon receiving necessary approvals from the Drugs Controller General of India.
Manufacturing
Manufacturing at company's India facility
Along with trials, Bharat Biotech will also be undertaking large-scale manufacturing of the vaccine at its GMP facility in Genome Valley, Hyderabad. "We are proud to collaborate on this innovative vaccine," said Dr. Krishna Ella, the Chairman and MD of Bharat Biotech. "We envision that we will scale this vaccine to 1 billion doses, translating to 1 billion individuals vaccinated receiving a single-dose regimen."
Advantage
Nasal vaccine expected to be more effective, easier to administer
A single dose nasal vaccine will be easier to administer, which will enable a broader reach while reducing the cost of medical consumables such as syringes. Precision Virologics Interim CEO David T. Curiel says that "it also prevents the spread of the disease by directly targeting the cells that line the nose and throat. Most other vaccine candidates currently under development can't do that."
COVAXIN
Bharat Biotech is also making its own homegrown COVID-19 vaccine
The licensing agreement comes as Bharat Biotech continues to work on its own, homegrown COVID-19 vaccine dubbed COVAXIN. The shot has been proven safe and effective in pre-clinical animal trials, and now, it is moving through the Phase-2 of human trials at multiple sites. The third phase is slated to begin sometime next month, according to recent reports.