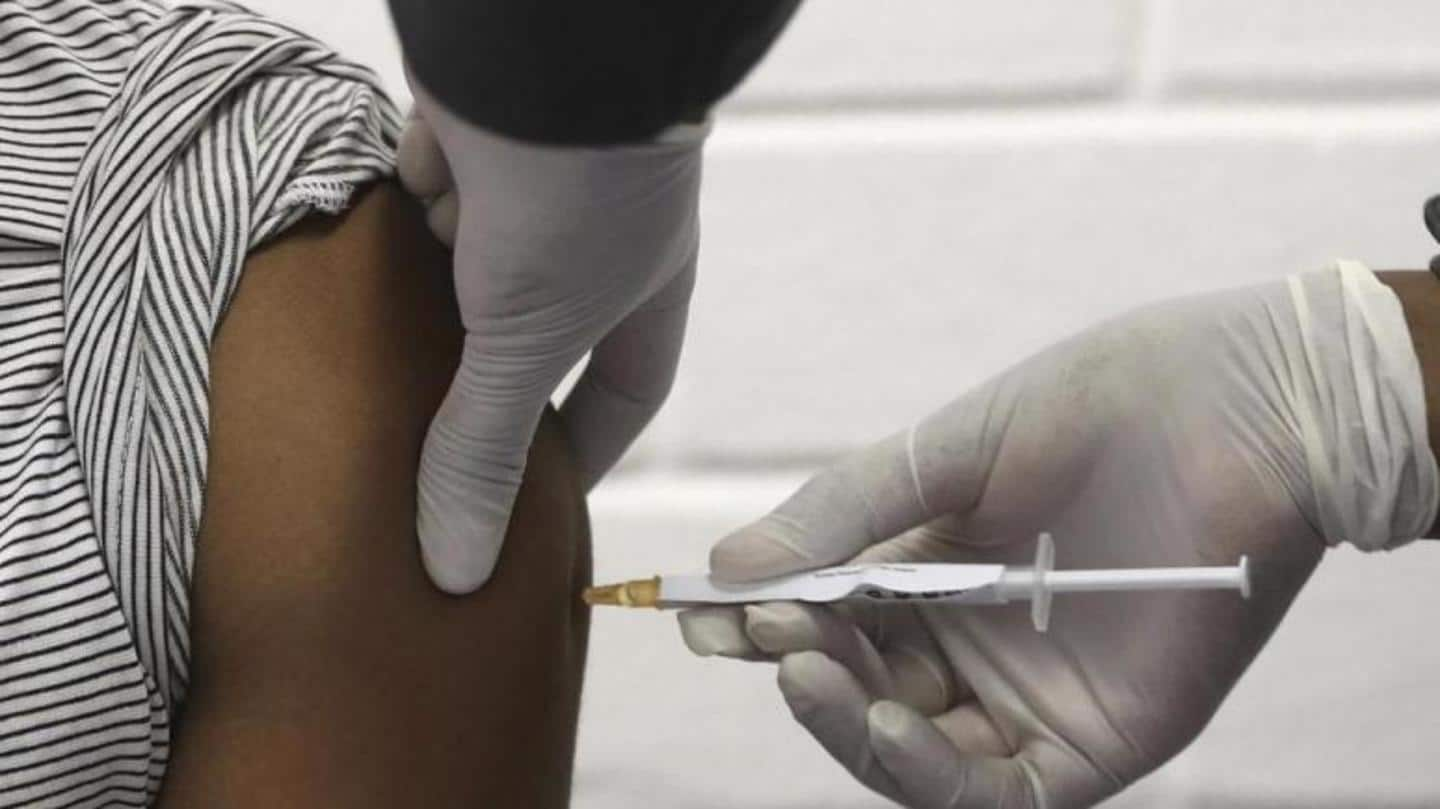
India's neighbors train experts for clinical trials of COVID-19 vaccine
What's the story
India's friendly neighboring nations trained over 100 experts to strengthen capacities in their countries for facilitating phase-3 clinical trials of the Indian COVID-19 vaccine, a senior official of the Department of Biotechnology said. The partnership for advancing clinical trials (PACT) initiative is being carried out in association with the Ministry of External Affairs, informed Alka Sharma, an advisor with the Department of Biotechnology.
Quote
Who is in charge of the initiative?
Addressing a webinar organized by the Confederation of Indian Industry, Sharma said, "The PACT is under the initiative of the Department of Biotechnology and the Ministry of External Affairs to strengthen capacities for facilitating phase-3 clinical trials of Indian COVID vaccine in friendly neighboring countries."
Participation
Which nations took part in the initiative?
Under this initiative, so far two training modules have been successfully completed. "More than 100 participants from Afghanistan, Bhutan, Maldives, Mauritius, Nepal, Sri Lanka took part in this training program," she said. The plan is to have the next round of training program with more number of participants, and currently 30 groups are actively involved in the development of the coronavirus vaccine, she informed.
Details
How are the vaccines coming along?
Six vaccines are in various stages of trials and four are being indigenously developed. Trials by Bharat Biotech are in phase-3, Zydus Cadila in phase-2, Biological E in phase-1. Gennova Biopharmaceuticals has got the nod for human trials I, II. Serum Institute of India and Dr. Reddy's Laboratories are in phase 2 and 3 clinical trials of the Oxford-Astrazeneca and Sputnik V vaccines, respectively.