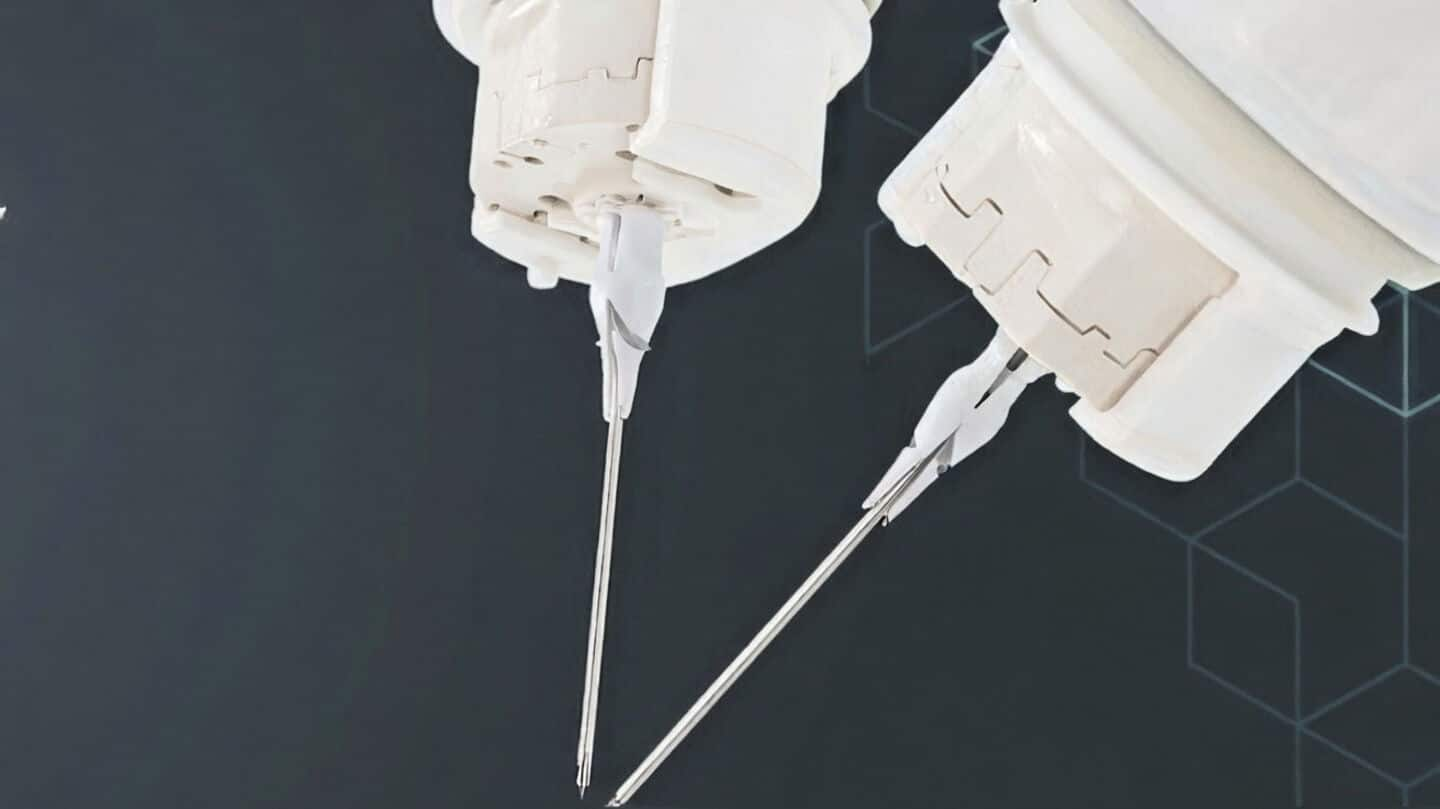
Medical Microinstruments gets FDA IDE for Symani Alzheimer's trial
Alzheimer's treatment might be getting a major upgrade:
MMI (Medical Microinstruments Inc.) just got FDA clearance to begin human trials under an Investigational Device Exemption (IDE) to test out tiny surgical robots instead of the usual meds.
Their REMIND trial will use the Symani Surgical System to see if robotic precision can help tackle neurodegenerative diseases like Alzheimer's.
REMIND trial targets deep cervical nodes
This study is all about targeting deep cervical lymph nodes, aiming to restore the brain's waste-clearing pathways and remove harmful proteins linked to Alzheimer's.
The surgery is super delicate because these vessels are tiny.
Over 30 days, researchers will track patient safety, and over six months, brain scans, and any changes in memory or thinking skills.
MMI's CEO calls it a new era for using robotics against tough diseases like Alzheimer's.